Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis
What is SDS-PAGE?
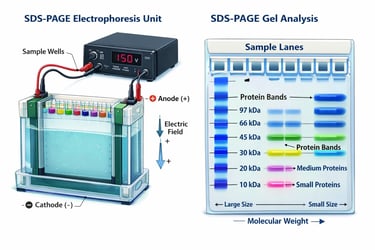
SDS-PAGE is a widely used technique to separate proteins based on their size. It is commonly used to analyze complex protein mixtures, check protein purity, and study protein subunits.
Principle of SDS-PAGE
SDS-PAGE is based on the principle of electrophoresis, where charged molecules move in an electric field.
Proteins normally differ in charge, size, and shape, which can affect how they migrate. However, in SDS-PAGE:
👉SDS unfolds proteins and gives them a uniform negative charge
👉This removes differences due to shape and charge
As a result, proteins move through a polyacrylamide gel, which acts like a mesh:
👉Smaller proteins move faster
👉Larger proteins move more slowly
🔹The gel itself is stable and has tiny pores that control movement, allowing proteins to be separated mainly based on their size (molecular weight).
What makes SDS-PAGE effective?
👉Uniform charge: SDS removes differences in protein charge and shape
👉Gel structure: The polyacrylamide gel controls movement based on size
👉High resolution: Can separate proteins with small size differences
Why is SDS-PAGE important?
👉Protein analysis: Check size and purity of proteins
👉Molecular biology: Study protein expression and structure
👉Biotechnology: Monitor recombinant protein production
👉Medical research: Detect protein-level changes in diseases
👉Foundation for other techniques: Used before methods like Western blot
To avoid confusion, it’s useful to compare SDS-PAGE with similar methods:
🔹SDS-PAGE: Proteins are unfolded → separated by size only
🔹Native PAGE: Proteins remain in natural form → separation by size, shape, and charge
🔹2D-PAGE: A more advanced method → first separates by charge, then by size (using SDS-PAGE)
Understanding Related Techniques (Quick View)
Xplore More